|
Until now, the accepted hypothesis was that Earth formed from dry materials, and that its water was delivered by celestial bodies that formed further from the sun: hydrated meteorites, such as carbonaceous chondrites, or comets – although this last hypothesis was recently thwarted by the ESA space probe Rosetta. Jon Taylor/Flickr, and Laurette Piani and Christine Fieni/MNHN, CC BY-SA Right: the Sahara 97096 meteorite, an enstatite chondrite with no hydrated minerals that formed in the inner part of the solar system (70 g). The Murchison meteorite, a carbonaceous chondrite containing hydrated minerals and organic components that formed in the outer part of the solar system (0.46 g). Contrastingly, ordinary and enstatite chondrites formed closer to the Sun where water was gaseous and was incorporated in large amounts into rocks: like the rocky planets, ordinary and enstatite chondrites are considered to be “dry”. The carbonaceous chondrites for instance formed far enough away from the Sun to initially contain water ice (all of which has since been incorporated in hydrated minerals through hydrothermal alteration). They are good witnesses of the first millions of years of the solar system. Some meteorites, called chondrites, come from small asteroids that, unlike the planets, have not geologically evolved since their formation. Laurette Piani The prevalent hypothesis: hydrogen delivered to Earth by hydrated asteroids At the low pressures of the interplanetary medium, the incorporation of water into planetary bodies depends on the surrounding temperature: above -184 degrees Fahrenheit, water is in its vapour form and does not agglomerate with other solids. The solar system began as a cloud of gas and dust, from which the planets and planetary bodies formed by the agglomeration of dust. So why does the Earth now have so much water, both in its mantle and on its surface? However, Earth and the other rocky planets (Mercury, Venus, and Mars) formed near the Sun, where it was too hot for water to incorporate into rock as ice: it just would have evaporated. 
In the early solar system, there was a lot of hydrogen, mainly in the form of dihydrogen gas (H 2), or bonded with oxygen atoms to form water (H 2O). 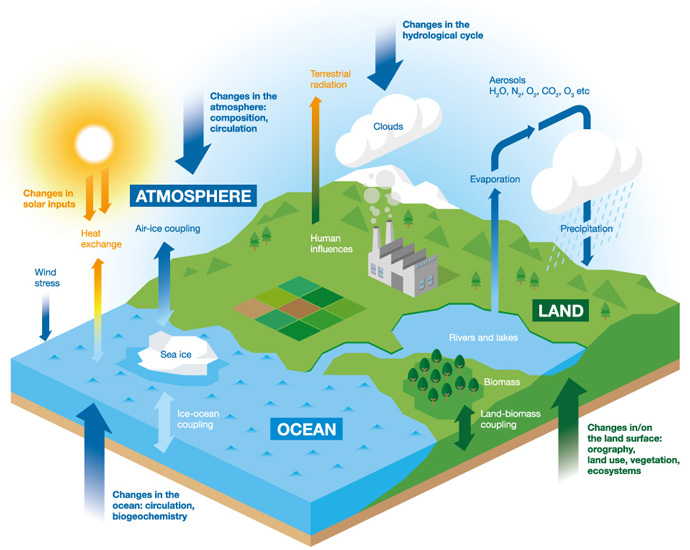
While water represents less than 0.5% of the mass of the Earth, it is key to the evolution of the planet itself and to life at its surface. 
This hydrogen can bond with surrounding oxygen to form water at the appropriate temperature and pressure conditions. Liquid water covers more than 70% of Earth’s surface, with about about 95.6% of it in oceans and seas, and the remaining 4% in glaciers, ice caps, groundwater, lakes, rivers, soil humidity, and the atmosphere.īut most of Earth’s water is deep underground: between one and ten times the volume of the oceans are contained in the mantle.Īt the surface of the Earth, “water” means two hydrogens for each oxygen (H 20), whereas what we call “water” in the mantle corresponds to hydrogen incorporated in minerals, magmas and fluids. Howard Perlman, USGS Jack Cook, Woods Hole Oceanographic Institution Adam Nieman, Author provided (no reuse) All surface water on Earth contained in a 1,300-km diameter sphere, that is, the size of Germany from north to south.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
